Novavax COVID-19 vaccine: Serum Institute to start trial next month in India
US drug developer Novavax Inc said on Tuesday it was doubling its potential COVID-19 vaccine manufacturing capacity to two billion doses annually
Union health minister Harsh Vardhan on Thursday said that COVID-19 vaccine may be available in India by first half of 2021
Astrazeneca also signed a deal with Serum Institute of India to manufacture Oxford COVID-19 vaccine (MINT_PRINT)
Serum Institute of India, the world's largest producer of vaccines, will begin the trial of Novavax COVID-19 vaccine in October, minister of state Ashwini Kumar Choubey informed the Lok Sabha on Friday.
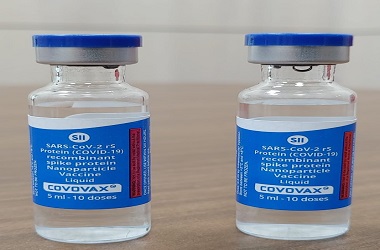
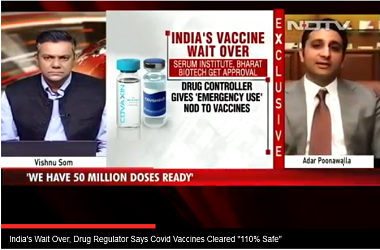
US drug developer Novavax Inc said on Tuesday it was doubling its potential COVID-19 vaccine manufacturing capacity to two billion doses annually under an agreement with Serum Institute of India. As part of the expanded agreement, Serum Institute will also manufacture the antigen component of the vaccine.
In August, Novavax signed a deal with Pune-based Serum Institute, to produce a minimum of one billion doses of its vaccine candidate, when approved, for low- and middle-income countries and India.
Dubbed NVX‑CoV2373, Novavax's vaccine is currently in mid-stage trials. The early-stage study showed it produced high levels of antibodies against the novel coronavirus.
"The trill will be initiated in second half of October after the vaccine is manufactured by the Serum Institute," Choubey said in his written reply to Lok Sabha. The trial is led by Indian Council of Medical Research-National AIDS Research Institute (NARI), Pune, Choubey added.
Commenting on the deal, Stanley C. Erck, president and chief executive officer of Novavax, " The agreement with Serum Institute enhances Novavax’ commitment to equitable global delivery of our COVID-19 vaccine. With this arrangement, we have now put in place a global supply chain that includes the recently acquired Praha Vaccines and partnerships with leading biologics manufacturers, enabling production on three continents."
British-Swedish firm Astrazeneca also signed a deal with Serum Institute of India to manufacture the COVID-19 vaccine developed by the University of Oxford. The advanced stage clinical trial of Oxford COVID-19 vaccine is presently going on in the country.
Union health minister Harsh Vardhan on Thursday said that COVID-19 vaccine may be available in India by first half of 2021. "We are not lagging behind anyone else in the whole world in our efforts to contribute towards the vaccine against COVID... In India we have about 7-8 vaccine candidates, three of them in the clinical trial phases and rest in the pre- clinical trials," Vardhan said earlier.
Source - liveMint
ABOUT US
OUR COMPANIES
CORPORATE SOCIAL RESPONSIBILITY
© Copyright 2026. Cyrus Poonawalla Group. All rights reserved.