Vaccine rollout will take two months after licensing, says Serum Institute's Poonawalla
The Serum Institute has taken on a vital role as the largest company licensed to manufacture the Oxford University-AstraZeneca coronavirus vaccine.
The company also has a deal with U.S.-based Novavax for their experimental COVID-19 vaccine but has not yet begun making it.

Post-licensing, it'll still take two months before large volumes rollout, says Adar Poonawalla, CEO of the Serum Institute of India, the world's largest manufacturer of vaccines.
The Serum Institute has taken on a vital role as the largest company licensed to manufacture the Oxford University-AstraZeneca coronavirus vaccine. It is increasing its production capacity by the end of 2021 to over 2.5 billion doses a year to cope with future disease outbreaks, Poonawalla said in a telephone interview with The Associated Press.
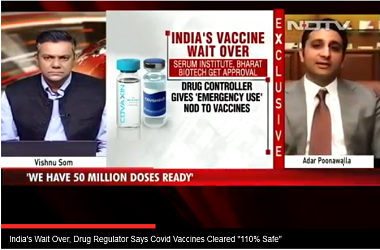
So far, the company has made 50 million doses of the Oxford University-AstraZeneca vaccine. It plans to make up to 100 million doses each month, starting next year, Poonawalla said.
The company also has a deal with U.S.-based Novavax for their experimental COVID-19 vaccine but has not yet begun making it. India has indicated that it wants 300 million doses by July. After that, the Serum Institute will be distributing vaccines to the world.
Poonawalla's company now has an annual capacity of 1.5 billion doses. That is more than the billion doses that China, home to the most vaccine makers in the world, says it manufactures in a year.
The coronavirus pandemic is a wake-up call for governments to invest more in health care, he added.
Looking beyond the pandemic, he said he anticipates more diseases jumping from animal to human hosts, driving huge demand for vaccines, so the extra capacity is likely to be useful in coming decades.
I think (the demand) is going to keep growing even more exponentially, compared to the last five or ten years, Poonawalla said Wednesday.
Loss of forests and human incursions into wild habitats create more opportunities for such pathogens to make the jump. That was true of COVID-19 and other illnesses such as SARS, MERS and HIV-AIDS.
Existing research indicates that India, with its high population density and biodiverse forests, is among the most likely places for the emergence of such new diseases.
For now, the Serum Institute is focused on the coronavirus. It has committed to supplying the Oxford University-AstraZeneca COVID-19 vaccine to developing nations through COVAX, an ambitious global initiative to distribute COVID-19 vaccines to countries worldwide.
Most of the vaccines that will be manufactured in the coming year have been reserved by richer nations, so Serum Institute's role will be pivotal, said Anna Marriott, a health policy adviser at Oxfam UK. Where else might the vaccine supply come from? she asked.
In April, before it was known whether any vaccine could work against the coronavirus, Serum bet on several vaccine candidates and invested in building its capacity.
Like many Indian companies, it is family-run, founded in 1966 by Poonawalla's father and its current chairman, Cyrus Poonawalla, and so had leeway to take that kind of risk.
The younger Poonawalla said the company faced a moral dilemma over waiting to be sure a vaccine might succeed and risking millions of lives.
But the pandemic has highlighted the power of vaccines since they generally are affordable and can help prevent expensive hospitalization, he said. Its a cheap and good solution to health care.
Countries spend too little on health care infrastructure, mistakenly viewing it as less important for the economy and political optics than investing in defense or space programs that highlight their growing might. That is particularly true, Poonawalla said, of developing nations like India, Whether it's hospitals, drug discovery (or) vaccines.
The pandemic should lead to greater support for research and health systems, I hope at least, he added
Source - CNBC-TV 18
ABOUT US
OUR COMPANIES
CORPORATE SOCIAL RESPONSIBILITY
© Copyright 2026. Cyrus Poonawalla Group. All rights reserved.