Pneumonia vaccine price drops dramatically for lower-income countries thanks to the Gavi pneumococcal Advance Market Commitment
New supply agreement between UNICEF and Serum Institute of India (SII) makes pneumococcal conjugate vaccine (PCV) available at US$2.00 per dose, a 43 per cent reduction from the Gavi price at the start of the Advance Market Commitment
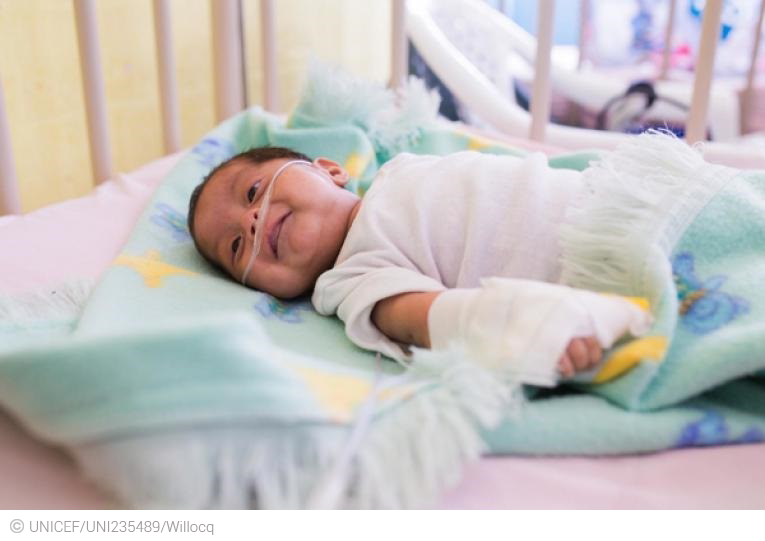
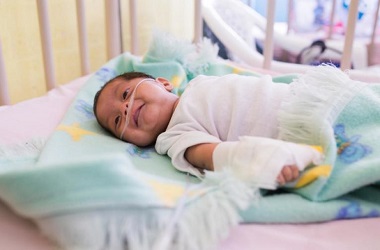
On October 22, 2019, at the National Hospital of Jalapa, Guatemala, Randy Hernández Estrada (6 months old) is being treated for pneumonia.
Geneva/ New York, 16 June 2020 – Lower-income countries across the world will now be able to access life-saving pneumococcal conjugate vaccines, which protect against the leading cause of pneumonia, for US$ 2 per dose, thanks to a new supply agreement between UNICEF, Gavi’s procurement partner, and the Serum Institute of India (SII). The new price represents a 43 per cent reduction from the Gavi price of US$3.50 at the start of the Advance Market Commitment (AMC).
The supply agreement is the eighth to take place under the Vaccine Alliance’s Advance Market Commitment (AMC) mechanism, and the first to include a developing country manufacturer. Under the agreement, the Indian manufacturer will provide ten million PCV doses to Gavi-supported countries each year for the next ten years.The pneumococcus bacterium is the leading cause of severe pneumonia and is a major cause of morbidity and mortality worldwide. Most of these deaths occur in lower-income countries and include a disproportionate number of children under the age of two.
The AMC, which is set to end this year, was launched by Gavi in 2009 – with the support of donors Italy, the United Kingdom, Canada, Russia, Norway and the Bill & Melinda Gates Foundation – to solve a clear example of market failure: complex vaccines like PCV would normally reach low-income countries, where the disease burden is often highest, ten to 15 years after their introduction in industrialised countries. The AMC mechanism incentivised research and development (R&D), particularly for vaccines suitable for developing country epidemiological contexts, and an expansion in manufacturing capacity to meet demand from lower-income countries while maintaining an affordable price per dose.
A decade later the results are clear: the PCV vaccine has now been introduced in 60 lower-income countries, where coverage rates, at 48%, are now higher than the global average of 47%. Estimates indicate that more than 225 million children will have been vaccinated, and that over 700,000 deaths will have been prevented by the end of 2020. In that time, the AMC has catalysed a steady decrease in PCV prices and thanks to this eighth supply agreement will be closing this year having facilitated the entry of a new manufacturer to the market as well as a record-setting low price for Gavi-supported countries that will result in an estimated millions of dollars in savings for both Gavi and lower income countries’ vaccine budgets.
“This is fantastic news, which will make this highly effective, lifesaving vaccine even more affordable for the world’s poorest countries, and is a testament to the success of Gavi’s Pneumococcal AMC mechanism, which as now achieved everything it set out to do,” said Aurelia Nguyen, Gavi’s Managing Director for Vaccines and Sustainability. “Thanks to this visionary model we have a healthy PCV market that is producing enough vaccines to supply both rich and poor countries and, as a result, hundreds of millions of children are now protected against one of the world’s deadliest diseases.”
“Pneumonia is the biggest single killer of children, claiming the life of a child every 39 seconds. By being able to provide this quality-assured pneumococcal conjugate vaccine at an such an affordable price, we can save millions of children’s lives,” said Etleva Kadilli, Director of UNICEF’s supply and procurement headquarters. “Our innovative approach has incentivized industry to bring this vaccine at scale to the market. The result means more countries – including middle-income countries – that have not yet introduced this vaccine into their routine immunization due to previously prohibitive pricing, will have an affordable option to accelerate access for all children."
“It is incredible to see that Gavi’s commitment to developing innovative mechanisms – and our donors’ commitment to supporting them – has had such a dramatic and positive impact on the lives of the most vulnerable children around the world,” said Marie-Ange Saraka-Yao, Gavi’s Managing Director for Resource Mobilisation, Private Sector Partnerships & Innovative Finance. “This successful mechanism will now be used as a model to help ensure the world’s poorest countries get access to COVID-19 vaccines at the same time as the wealthiest, making it integral to the global effort to defeat this pandemic.”
At last week’s Global Vaccine Summit, Gavi launched the Gavi Advance Market Commitment for COVID-19 Vaccines (Gavi Covax AMC), a new financing instrument aimed at incentivising vaccine manufacturers to produce sufficient quantities of eventual COVID-19 vaccines, and to ensure access for developing countries.
Source - UNICEF
ABOUT US
OUR COMPANIES
CORPORATE SOCIAL RESPONSIBILITY
© Copyright 2026. Cyrus Poonawalla Group. All rights reserved.