Oxford COVID-19 vaccine 95% effective if given 3 months apart, says SII's Adar Poonawalla
Serum Institute of India (SII) CEO Adar Poonawalla said on December 28, that the vaccine is likely to get an emergency-use licence from the UK Medicines and Healthcare products Regulatory Agency as well as the Indian regulator, Drugs Controller General of India (DCGI), by early January

Serum Institute of India (SII) CEO Adar Poonawalla
British-Swedish pharma giant AstraZeneca Plc will soon come out with data manifesting that the efficacy of its coronavirus vaccine rises to as much as 95 per cent if the two shots are parted by around 2-3 months, said Adar Poonawalla, Chief Executive Officer, Serum Institute of India (SII).
SII is AstraZeneca's manufacturing partner. Speaking at the launch of Serum Institute's pneumococcal conjugate vaccine, which is the first such indigenous vaccine, under the brand Pneumosil, Poonawalla said, "You'll be hearing some good news from the UK very soon... It would be a 90-95% effective vaccine if you just keep a two-to-three months' gap between dose 1 and dose 2. They will make that public with documentation."
His remarks follow a statement by AstraZeneca CEO Pascal Soriot, who said that the company has worked out a "winning formula to get efficacy up there with everybody else." Soriot said it in an interview with Britain's Sunday Times newspaper. Although he did not give out any details to support his claim, he informed that the data would be published at "some point."
Efficacy outcomes of AstraZeneca's vaccine, co-developed with Oxford University, triggered controversy after it claimed that a dosing error accelerated the effectiveness of the vaccine.
Admitting the same in the interview, Soriot said, "we would have preferred a simpler set of results." The British-Swedish pharma giant's clinical trials for its vaccine in the UK and Brazil comprised two distinct dosing regimens. A lower dosage demonstrated a greater efficacy of 90 per cent, while two full doses a month apart manifested a 62 per cent efficacy.
Meanwhile, Poonawalla said on Monday, December 28, that the vaccine is likely to get an emergency-use licence from the UK Medicines and Healthcare products Regulatory Agency as well as the Indian regulator, Drugs Controller General of India (DCGI), by early January.
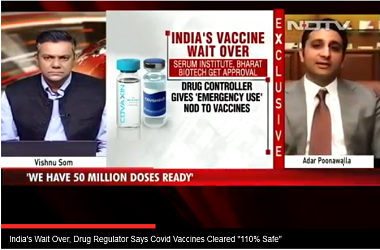
Presently, three drugmakers - Bharat Biotech, Pfizer and Serum Institute - have applied for emergency licences, which a subject expert panel established by the DCGI is likely to vet soon. SII and Bharat Biotech had earlier this month submitted more data, while Pfizer asked for more time to furnish its proposal.
Source - Business Today
ABOUT US
OUR COMPANIES
CORPORATE SOCIAL RESPONSIBILITY
© Copyright 2026. Cyrus Poonawalla Group. All rights reserved.