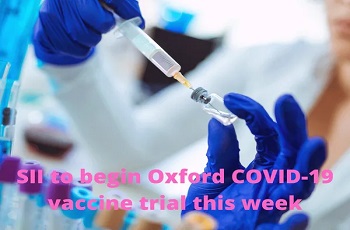
Serum Institute of India gets approval to start human trials for Oxford vaccine
Earlier this week, a committee of experts had deferred a decision on the request of SII to start trials and asked the company to amend its protocol for the clinical study. The subject expert committee (SEC) had recommended eight amendments to be made to the firm's proposal to conduct Phase-II and -III trials.
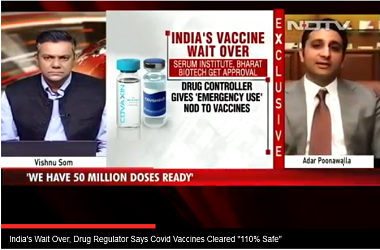
NEW DELHI: The Serum Institute of India (SII) got the approval on Friday to start human trials in India for the Covid-19 vaccine candidate developed by University of Oxford, after the company submitted a revised protocol to the drug regulatory authority, people in the know told ET.
On Wednesday, a committee of experts had deferred a decision on the request of SII to start trials and asked the company to amend its protocol for the clinical study. The subject expert committee (SEC) had recommended eight amendments to be made to the firm's proposal to conduct Phase-II and -III trials.
According to the minutes of Wednesday's meeting, the SEC had asked Serum to distribute clinical trial sites across India and demarcate the phase-II and Phase-III part of the protocol. ET has seen the minutes of the meeting.
As per the new proposal, 1,600 people will participate in the trials in over 20 cities spread across India.
A meeting of the SEC was called on Friday where it gave the approval to Serum's amended protocol, a government official said.
Serum has tied up with Swedish-British drug maker AstraZeneca to produce around a billion doses for low- and middle-income countries by early next year.
The group of experts had earlier asked the Pune-based company to also revise the dropout rate of 41% proposed in the study "to keep the rate at par with other vaccine studies".
"Immunogenicity shall be either primary or secondary objective," stated the minutes of the meeting. It added that cellular immunity - the ability of the body to develop cells that specifically target and protect against the virus -should be a part of the immunogenicity assessment, among other recommendations.
The committee had also said an interim analysis of the participants being studied for an immune response might be carried out "only after day 58, both for safety and immunogenicity".
Source - The Economic Times
ABOUT US
OUR COMPANIES
CORPORATE SOCIAL RESPONSIBILITY
© Copyright 2026. Cyrus Poonawalla Group. All rights reserved.